Krabbe Disease Treatment Market is progressing through gene therapy innovation and increased funding for rare disease care to improve outcomes.
ROCKVILLE , MD, UNITED STATES, May 4, 2026 /EINPresswire.com/ — Krabbe Disease Treatment Market valued at USD 2.88 Bn in 2024, projected to reach USD 5.03 Bn by 2032 at 7.22% CAGR – gene therapy breakthroughs and newborn screening expansion are transforming this rare neurological disorder’s therapeutic landscape.
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/272964/
Overview: Gene Therapy and Newborn Screening Redefine a USD 5.03 Billion Rare Disease Market
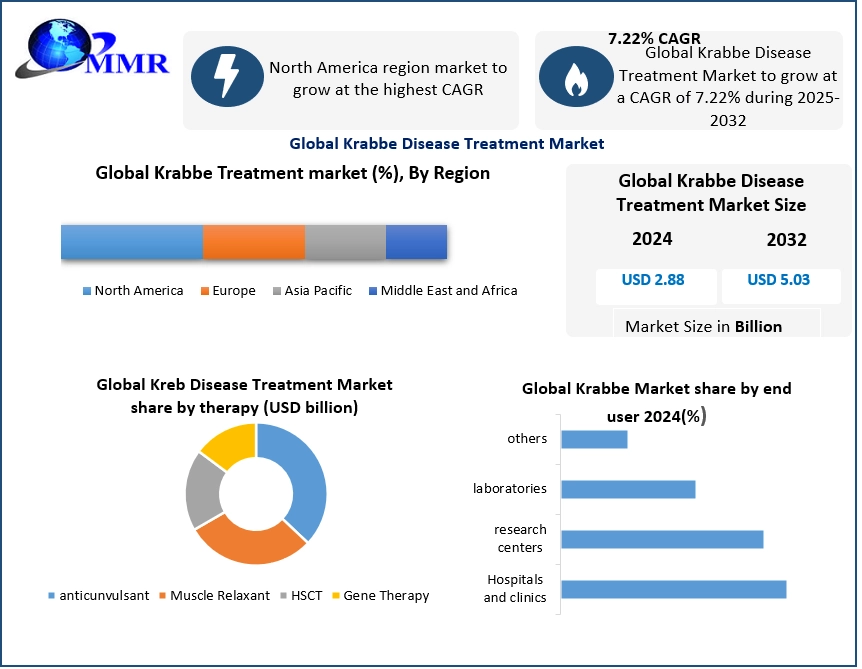
The Global Krabbe Disease Treatment Market size was valued at USD 2.88 billion in 2024 and is projected to reach USD 5.03 billion by 2032 at a CAGR of 7.22%. Growth is driven by expanding newborn screening programs and advancements in gene therapy targeting GALC deficiency. Increasing orphan drug incentives and clinical pipeline progress are improving early diagnosis and treatment outcomes, accelerating adoption and supporting krabbe disease treatment market size growth globally.
Market Dynamics: Drivers, Restraints & Opportunities
Drivers: Orphan Drug Designations, Gene Therapy Investment, and Newborn Screening Expansion Accelerate Market Growth
FDA orphan drug and rare pediatric disease designations – granted to pipeline candidates including Passage Bio’s PBKR03 – provide market exclusivity and R&D tax incentives that are structurally accelerating pharmaceutical investment into Krabbe disease therapeutics. Parallel expansion of newborn screening programs across U.S. states enables presymptomatic HSCT, the only intervention proven to slow disease progression, directly expanding the treatable patient population and driving commercial demand for specialist treatments.
Restraints: Limited Patient Pool, Low Diagnostic Awareness, and Trial Recruitment Barriers Constrain Growth Velocity
Krabbe disease affects approximately 1 in 100,000 live births in the United States, severely limiting clinical trial cohorts and slowing regulatory evidence generation. Low awareness among general practitioners frequently results in diagnostic delays, reducing the window for effective early intervention. The COVID-19 pandemic further disrupted rare disease research timelines, creating residual setbacks in therapeutic development pipelines that continue to temper near-term market velocity.
Opportunities: AAV Gene Therapy Pipelines, FDA Breakthrough Pathways, and Pfizer’s Rare Disease Investment Create Significant Market Upside
Adeno-associated viral vector gene therapy targeting the underlying GALC mutation represents the most commercially transformative opportunity in the Krabbe disease treatment landscape. Pfizer’s acquisition of Biohaven Pharmaceuticals signals major pharma’s deepening commitment to rare neurological disorders. FDA Breakthrough Therapy and Fast Track designations are accelerating review timelines, compressing time-to-market for clinical-stage Krabbe therapies and driving substantial near-term investment into the sector.
Key Market Trends: Gene Therapy, HSCT Expansion, and Screening Programs Driving 2025–2032 Krabbe Disease Treatment Landscape
Forge Biologics’ FBX-101 REKLAIM Trial Targets Both Infantile and Late-Onset Krabbe Disease
Forge Biologics’ FBX-101, an AAV-based gene therapy, is being evaluated in the REKLAIM trial across infantile and late-onset Krabbe patients. This dual-cohort approach could expand treatment coverage and position FBX-101 as a leading gene therapy candidate.
Newborn Screening Mandates Are Expanding the Presymptomatic HSCT Treatment Window
Expanded newborn screening in multiple U.S. states is increasing early detection of Krabbe disease. Presymptomatic HSCT remains the most effective treatment, improving survival outcomes. Wider screening is expanding the eligible patient pool and driving growth in specialized treatment centers.
Pfizer’s Biohaven Acquisition Signals Major Pharma’s Rare CNS Disease Commitment
Pfizer’s Biohaven acquisition signals rising big pharma focus on rare neurological disorders, including Krabbe disease. The move is attracting institutional investment into early-stage pipelines and accelerating the transition from academic research to commercial drug development across emerging therapies.
Krabbe Disease Treatment Market Segmentation: Anticonvulsants Lead as Gene Therapy Becomes Fastest-Growing Segment
Anticonvulsants lead the Krabbe disease treatment market size, supporting seizure management, while HSCT remains the standard for presymptomatic infants and gene therapy is the fastest-growing segment. Infantile cases account for 85–90% of demand. The U.S. leads globally due to strong screening programs and specialized transplant centers driving advanced treatment adoption.
By Disease Type
Infantile
Late-onset
By Treatment Insight
Anticonvulsants
Muscle relaxants
HSCT
Gene Therapy
By End Users
Hospitals and Clinics
Research Centers
Laboratories
Others
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/272964/
Regional Insights: North America Commands Market Leadership While Asia Pacific Emerges as the Next Frontier
North America: Advanced HSCT Infrastructure, Newborn Screening Mandates, and Gene Therapy Pipeline Anchor Market Dominance
North America leads the Krabbe disease treatment market, supported by advanced rare disease infrastructure and expanding newborn screening programs. U.S. FDA orphan drug incentives are accelerating gene therapy development by key players. Growing specialized treatment capacity in the U.S., Canada, and Mexico further strengthens regional leadership and market growth through 2032.
Europe: High Late-Onset Prevalence, EURODIS Infrastructure, and Centralized Rare Disease Policy Drive Second-Largest Market
Europe ranks second in the Krabbe disease treatment market, supported by strong rare disease networks like EURODIS. Higher late-onset prevalence and advanced research ecosystems drive demand. Israel’s high incidence rates strengthen its role in clinical research, while companies like Orchard Therapeutics support regional gene therapy development and market growth through 2032.
Key Recent Developments: Krabbe Disease Treatment Market (2021–2025)
Forge Biologics (2023): Forge Biologics launched the REKLAIM Phase 1/2 clinical trial for FBX-101 – an AAV gene therapy targeting GALC mutation – enrolling both infantile and late-onset Krabbe patients, making it the broadest-enrollment gene therapy trial in Krabbe disease history.
Passage Bio (2022): Passage Bio’s PBKR03 received FDA Orphan Drug and Rare Pediatric Disease designations, advancing into Phase 1/2 trials. Successful Phase 1 safety data opened a pivotal trial pathway, with Rare Pediatric Disease designation conferring a Priority Review Voucher upon approval.
University of Pittsburgh / UPMC (2023): University of Pittsburgh researchers received federal NIH funding to advance newborn screening methodologies for Krabbe disease – improving enzyme assay sensitivity and specificity to reduce false positives, directly expanding the presymptomatic HSCT-eligible infant population.
Pfizer — Biohaven Acquisition (2022): Pfizer’s USD 11.6 billion acquisition of Biohaven Pharmaceuticals signaled major pharma’s accelerating commitment to rare CNS disorders, catalyzing broader institutional investment into orphan neurological markets including Krabbe disease gene therapy pipelines.
Key Players:
Pfizer
Novartis
Sanofi
Takeda Pharmaceutical
Johnson & Johnson
Roche
AstraZeneca
Biogen
Regeneron Pharmaceuticals
Vertex Pharmaceuticals
Bluebird Bio
Sarepta Therapeutics
Ultragenyx Pharmaceutical
Orchard Therapeutics
Rocket Pharmaceuticals
Get access to the full description of the report @ https://www.maximizemarketresearch.com/market-report/krabbe-disease-treatment-market/272964/
Competitive Landscape of the Krabbe Disease Treatment Market
The Krabbe Disease Treatment Market is defined by a bifurcated competitive structure: established pharmaceutical players managing symptom-based anticonvulsant demand, and a dynamic gene therapy vanguard led by Forge Biologics, Passage Bio, and Orchard Therapeutics. Forge’s REKLAIM trial and Passage Bio’s PBKR03 represent the two most advanced curative-intent pipeline assets. Pfizer, Novartis, and Biogen maintain rare neurological disorder portfolios with potential adjacency to Krabbe, while competitive positioning through 2032 will be decisively shaped by Phase 2/3 clinical readouts and FDA review timelines.
Analyst Perspective: From Symptom Management to Genetic Cure — Krabbe Disease Treatment Is Entering a New Era
For decades, Krabbe disease treatment meant managing deterioration – not preventing it. Gene therapy is fundamentally altering that reality. AAV-delivered GALC replacement, combined with presymptomatic HSCT enabled by mandatory newborn screening, represents the first genuine pathway toward disease arrest. The commercial opportunity between 2025 and 2032 belongs to those who move fastest from clinical proof-of-concept to regulatory approval – the window is narrow, but the therapeutic and market impact will be transformative. — Maximize Market Research
FAQs:
What is the global Krabbe Disease Treatment Market size and forecast?
The Krabbe disease treatment market size was USD 2.88 billion in 2024 and is projected to reach USD 5.03 billion by 2032 at a 7.22% CAGR, driven by gene therapy, orphan drug incentives, HSCT expansion, and newborn screening growth.
What is the most effective current treatment for Krabbe disease?
Hematopoietic stem cell transplantation (HSCT) is the most effective treatment for presymptomatic infantile Krabbe disease, improving survival outcomes. Anticonvulsants remain the dominant segment for managing symptoms across both infantile and late-onset cases globally..
Which gene therapy candidates are furthest in Krabbe disease clinical development?
Forge Biologics’ FBX-101 and Passage Bio’s PBKR03 are leading clinical-stage gene therapies. Both use AAV vectors to deliver functional GALC genes, aiming to slow or halt neurological progression in Krabbe disease.
What are the biggest growth opportunities in the Krabbe disease treatment sector?
Key growth opportunities include FDA Breakthrough and Fast Track designations for gene therapies, Pfizer’s investment in rare CNS disorders, expanding newborn screening programs, and rising unmet need in late-onset Krabbe disease treatment.
Related Reports:
Global Scanning Electron Microscopes Market: https://www.maximizemarketresearch.com/market-report/scanning-electron-microscopes-market/187558/
Scanning Electron Microscopes Market by Type (Conventional SEM, Field Emission SEM, Desktop SEM), Application (Semiconductors, Life Sciences, Materials), End-User (Research Institutes, Industries) and Region – Global Forecast to 2032
Global Bioinformatics Market: https://www.maximizemarketresearch.com/market-report/bioinformatics-market/207971/
Bioinformatics Market by Product & Services (Knowledge Management Tools, Platforms, Services), Application (Genomics, Proteomics, Transcriptomics, Metabolomics, Chemoinformatics), End-User and Region – Global Forecast to 2030
Top Reports:
Photocatalytic Coatings Market
Global Veterinary Reference Laboratory Market
About Maximize Market Research
Maximize Market Research is a leading market research and business consulting firm delivering specialized intelligence across rare disease therapeutics, medical biotechnology, and global healthcare markets. Our rare disease research capabilities support pharmaceutical developers, hospital systems, and institutional investors with actionable insights aligned to regulatory timelines, clinical pipeline milestones, and evolving treatment paradigms.
Lumawant Godage
MAXIMIZE MARKET RESEARCH PVT. LTD.
+ +91 96073 65656
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()
Media gallery